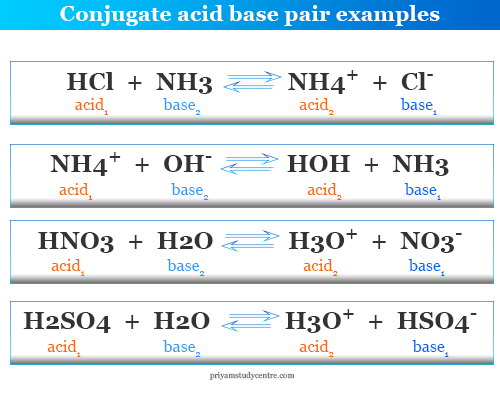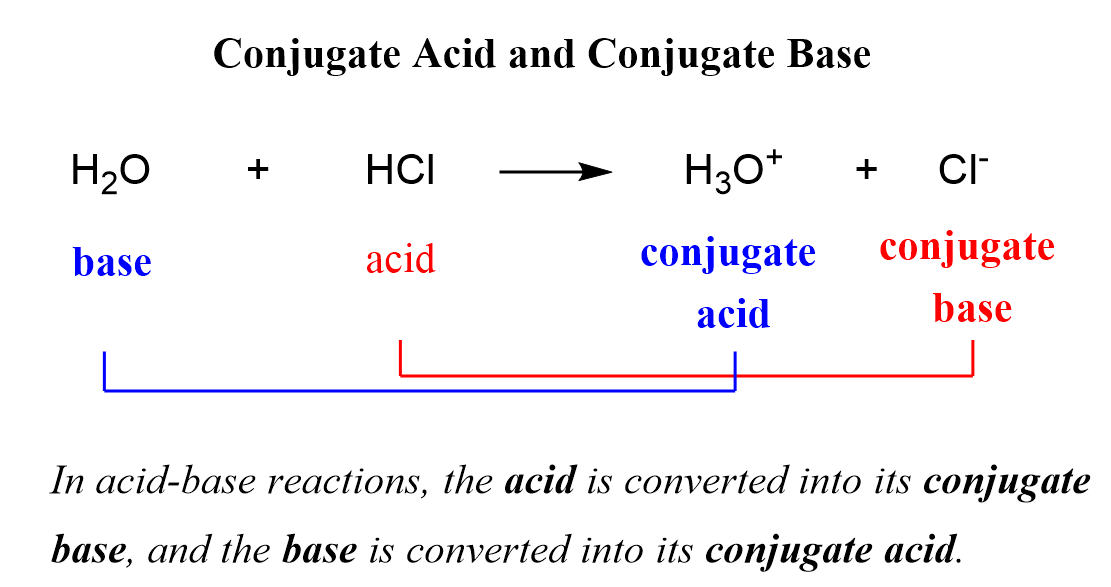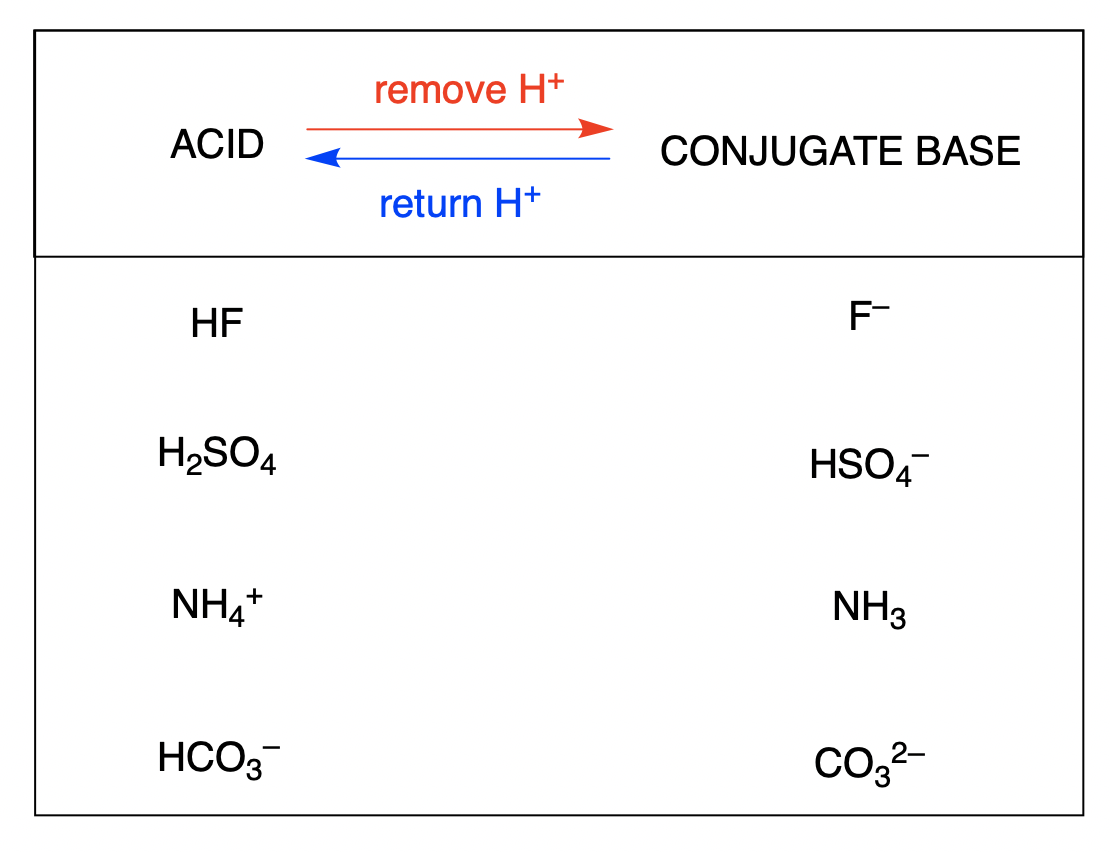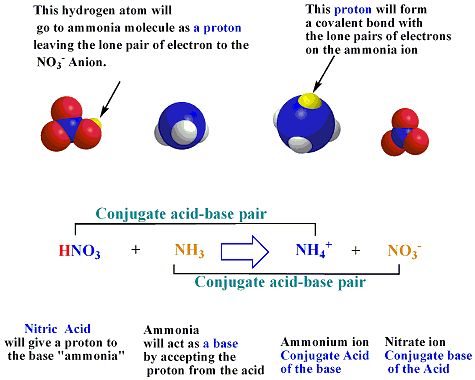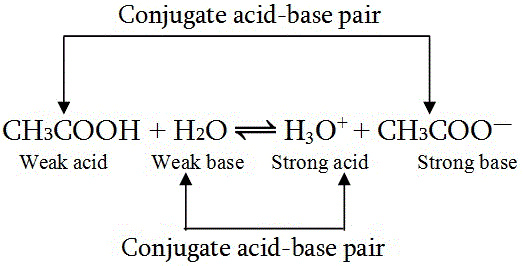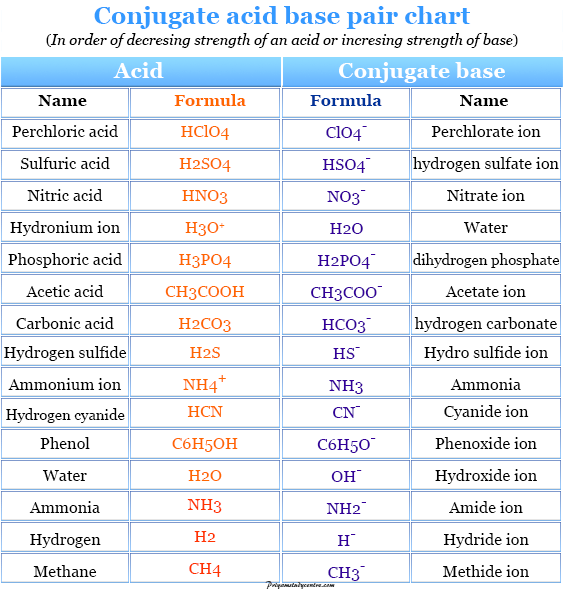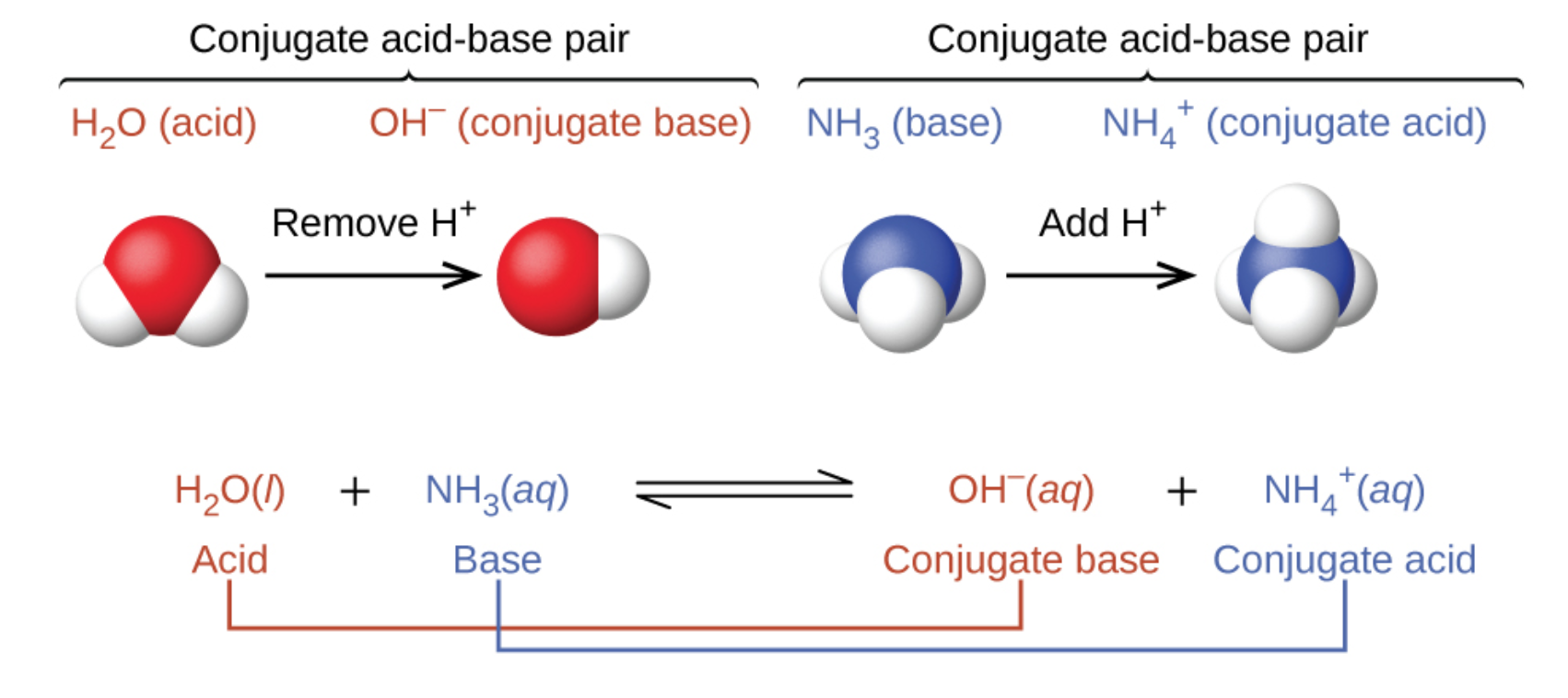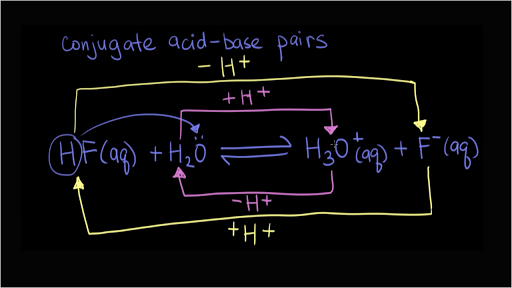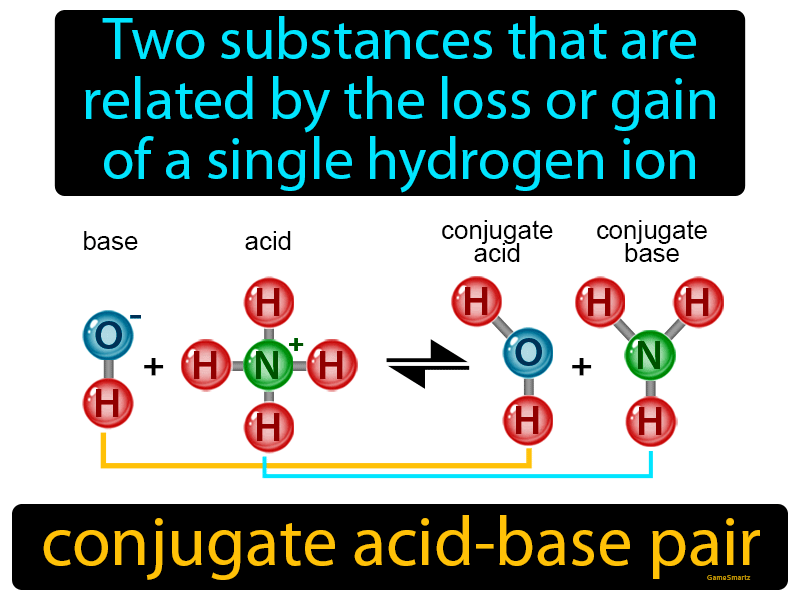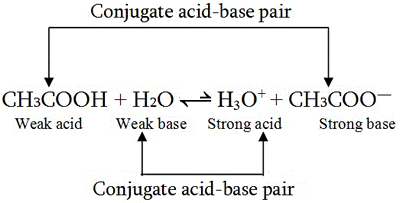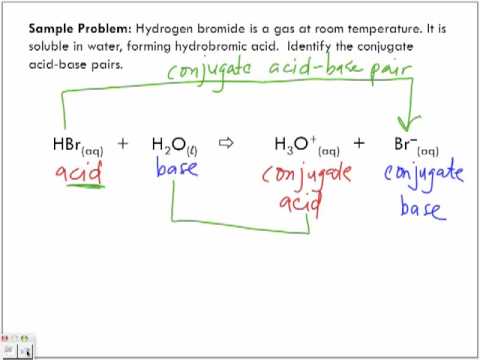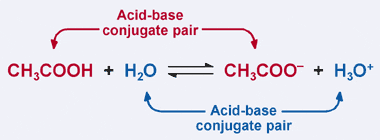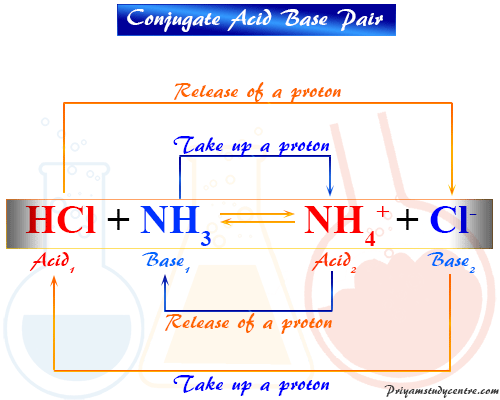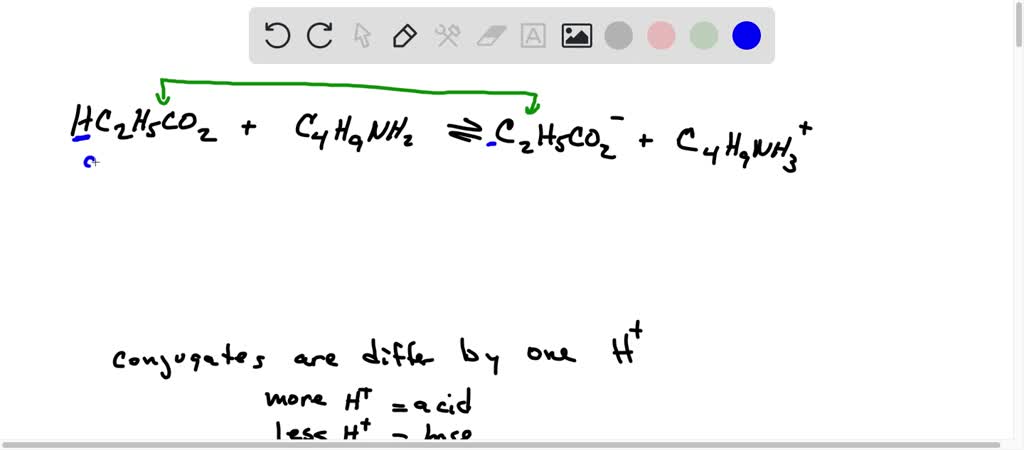
SOLVED: Using the Brønsted-Lowry definition of acids and bases, identify the conjugate acid-base pairs in this reversible reaction. (Omit states-of-matter from your answer.) HC2H5CO2(aq) + C4H9NH2(aq) C2H5CO2−(aq) + C4H9NH3+(aq) Acid? Conjugate base?

Identify the conjugate acid-base pairs for the reaction (with the acid written first). CN- + H2O = HCN + OH- |CN- / HCN |HCN / CN- |OH- / H2O |H2O / OH- | Homework.Study.com