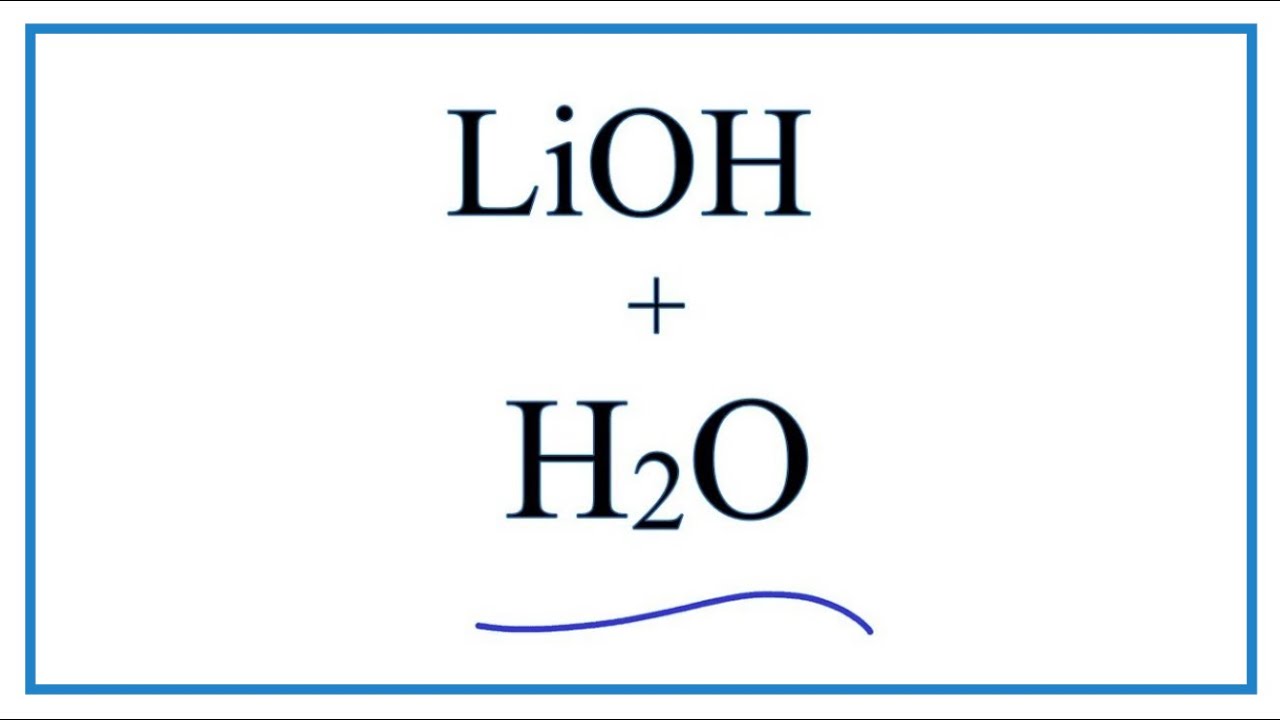
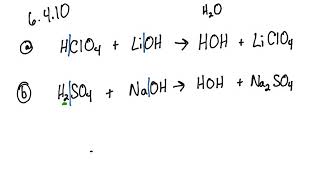Question Video: Calculating the Concentration of Lithium Hydroxide in Millimolars Via Titration with Sulfuric Acid | Nagwa

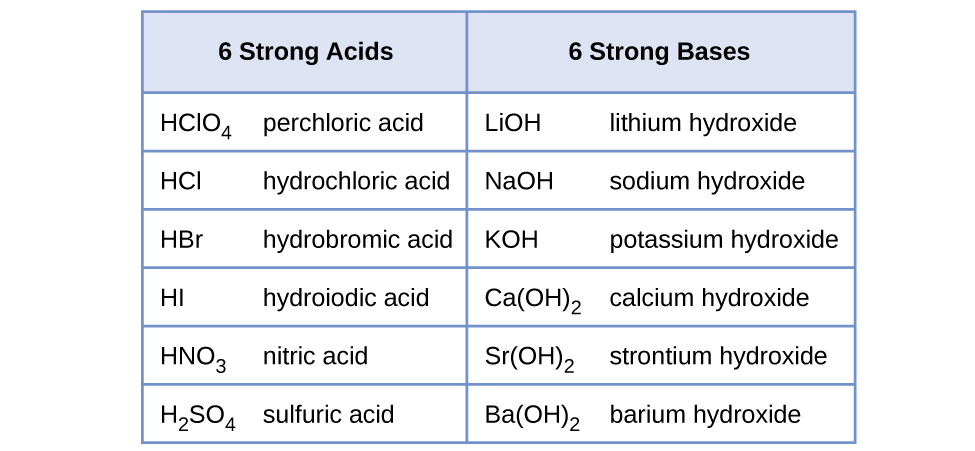
PPT - STRONG BASES LiOH, NaOH, KOH, RbOH, CsOH, Ca(OH) 2 , Sr(OH) 2 , Ba(OH) 2 PowerPoint Presentation - ID:4763546

Titration curves for solutions with 1.0 × 10 −4 moles of LiOH·H 2 O and... | Download Scientific Diagram

SOLVED: QUESTION 7 Which of the following equations represents the acid-base neutralization reaction between H3PO4 ad LiOH? Note that coefficients for balancing the final equation are not included: H3PO4 + LiOH LizPO4 -

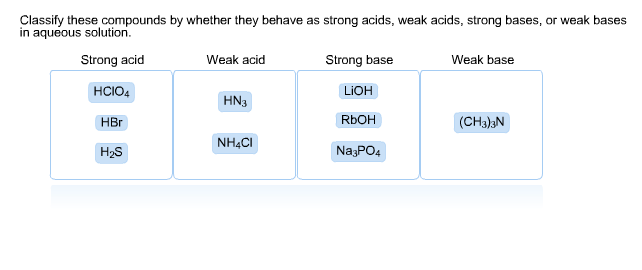
How to Identify the Major Species in a Mixture of Weak and Strong Acids or Bases | Chemistry | Study.com